Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters

Research — April 1, 2026
By Sarthak Tounk
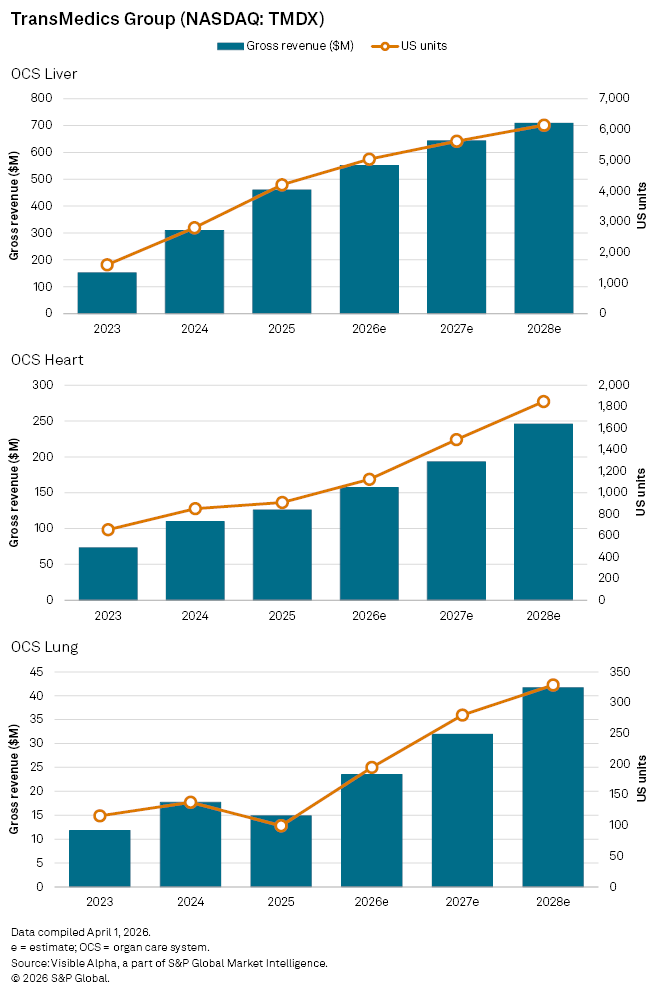
US organ transplant therapy provider TransMedics Group Inc. (NASDAQ: TMDX) is advancing its clinical pipeline, with its ENHANCE Heart and DENOVO Lung trials, both cleared by the U.S. Food and Drug Administration and now entering early activation and patient enrolment. Analysts expect these programs to begin translating into stronger growth across the company’s Organ Care System (OCS) portfolio from 2026.
Visible Alpha consensus points to OCS Heart revenues reaching $157 million in 2026, an increase of 25% year-on-year, supported by US unit volumes rising to 1,124 from 909 in 2025.
Growth in the smaller OCS Lung segment is forecast to be more pronounced. Revenues are expected to rise 58% to $24 million in 2026, reversing a 16% decline last year, while US volumes are projected to nearly double to 194 units from 99.
OCS Liver, the company’s largest division, is set to remain the primary earnings driver. Analysts forecast revenues of $551 million in 2026, up 20%, with US volumes increasing to 5,029 from 4,193, reflecting continued penetration in what is currently the most established application of the platform.
Beyond its existing franchises, TransMedics is targeting expansion into kidney transplantation, the largest segment of the market and one marked by significant unmet need. The company is developing a next-generation warm perfusion system designed to support the entire donor-to-recipient pathway, with plans to enter FDA trials by early 2027. If successful, the move would materially broaden its addressable market and reinforce its positioning in the rapidly evolving field of organ preservation technology.
TransMedics is the only FDA-approved, portable platform offering warm perfusion for heart, lung and liver transplants, replacing passive cold storage and thereby reducing post-transplant complications.
This article was published by Visible Alpha, part of S&P Global Market Intelligence and not by S&P Global Ratings, which is a separately managed division of S&P Global.
Content Type
Products & Offerings
Segment
