Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Research — April 16, 2026
By Jueeli Kadam
Vertex Pharmaceuticals Inc. (NASDAQ: VRTX) has taken a significant step towards diversifying beyond its dominant cystic fibrosis franchise, after its experimental IgA nephropathy (IgAN) therapy, povetacicept, delivered positive Phase 3 results. The outcome positions the biotech for a potential accelerated approval as early as November.
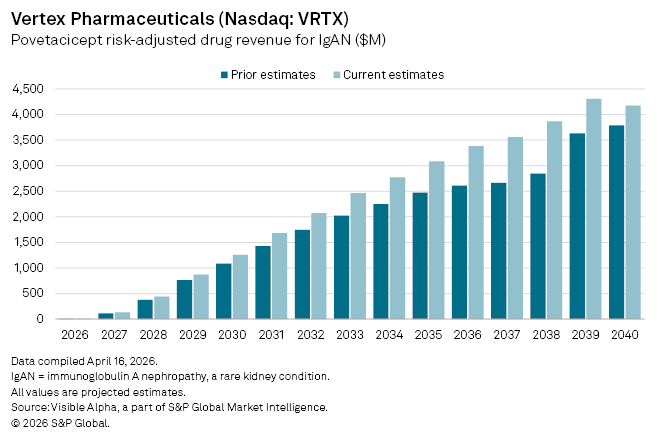
Analysts have responded swiftly, raising the drug’s probability of success to 80% from 70.86%, reflecting increased confidence in both regulatory prospects and commercial viability. The upgrade feeds directly into revenue expectations, with consensus estimates for risk-adjusted sales in IgAN climbing to $2.1 billion in 2026, up from $1.3 billion previously, contingent on approval later this year.
Near-term projections have also edged higher. First full-year sales are now seen at $132 million, compared with earlier estimates of $114 million. Longer term expectations have been revised meaningfully, with forecasts now pointing to blockbuster sales of $1.3 billion by 2030, versus $1 billion previously, before scaling further to peak global sales of $4.3 billion by 2039.
Vertex’s push into IgAN places it in direct competition with established players including Novartis AG (SIX: NOVN) and Otsuka Holdings Co. Ltd. (TYO: 4578), both of which already market approved therapies, as well as Vera Therapeutics Inc. (NASDAQ: VERA), which is advancing its own late-stage candidate. While the competitive landscape is intensifying, the scale of the opportunity is expanding rapidly.
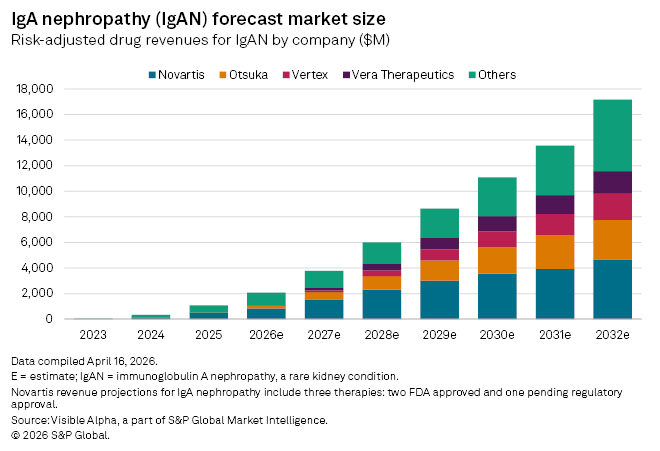
Data from the Visible Alpha Biopharma universe highlights that growth trajectory, with the global IgAN market expected to increase from $2.1 billion in 2026 to $17.2 billion by 2032, spanning 38 drugs across 20 companies.
This article was published by Visible Alpha, part of S&P Global Market Intelligence and not by S&P Global Ratings, which is a separately managed division of S&P Global.
Content Type
Products & Offerings
Segment

