Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Research — April 8, 2026

By Anjali Pandey
Novo Nordisk A/S (CPH: NOVO-B) has gained a first-mover advantage in the US market for next-generation insulin therapies after the US FDA approved Awiqli (insulin icodec-abae), the first once-weekly basal insulin for adults with type 2 diabetes. Awiqli is already available in parts of EU and other international markets, though labelling varies, and is expected to launch commercially in the US by the second half of the year.
Despite this early lead, analyst attention is increasingly on Eli Lilly and Co. (NYSE: LLY) and its rival once-weekly insulin candidate, efsitora alfa, which remains under regulatory review across the US, Europe and Japan. Visible Alpha consensus forecasts suggest Lilly’s entrant could ultimately outpace Awiqli commercially, reflecting both its competitive positioning and Lilly’s strong hold in the US diabetes and obesity market.
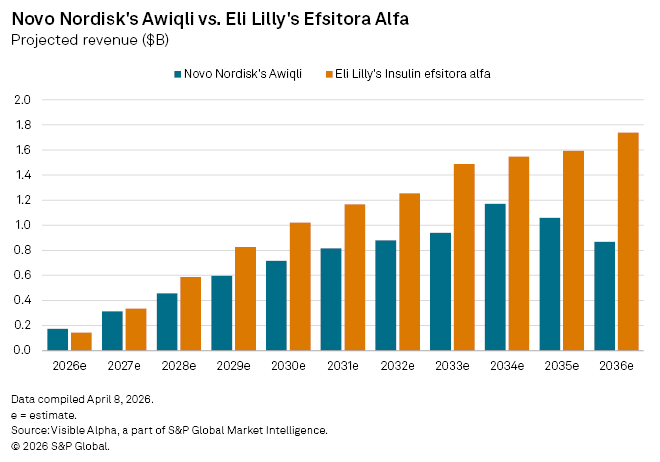
Visible Alpha estimates point to Awiqli generating around $174 million in revenue in 2026, rising from $65 million in 2025 (prior to its US approval), and reaching approximately $311 million by 2027. By comparison, efsitora alfa, assuming approval later this year, is forecast to deliver $144 million in risk-adjusted sales in 2026, climbing to $335 million in 2027.
Longer term, the divergence becomes more pronounced. Awiqli’s peak global sales are projected at about $1.2 billion by 2034. Efsitora alfa, by contrast, is expected to reach blockbuster status earlier, potentially exceeding $1 billion in annual sales by 2030 and peaking at roughly $2.5 billion by 2037.
This article was published by Visible Alpha, part of S&P Global Market Intelligence and not by S&P Global Ratings, which is a separately managed division of S&P Global.
Content Type
Products & Offerings
Segment
