Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Research — February 20, 2026
By Jueeli Kadam
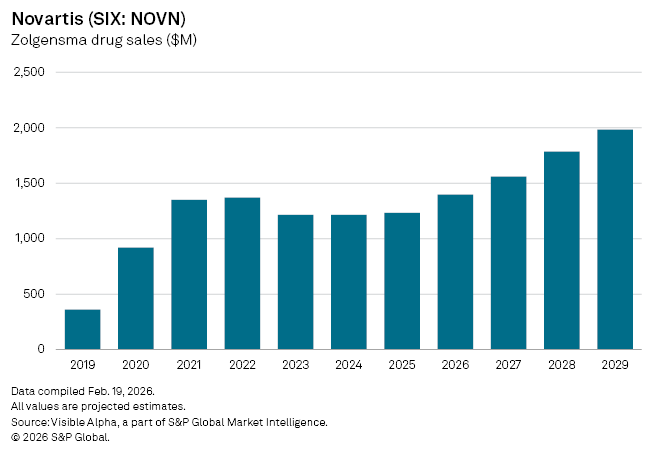
Swiss pharma giant Novartis AG (SIX: NOVN) secured a fresh foothold in the spinal muscular atrophy (SMA) market after US FDA approved Itvisma, the latest iteration of its gene therapy Zolgensma, for patients aged 2 years and older. The FDA cleared the treatment in November 2025 for patients 2 years and older with a confirmed mutation in the SMN1 gene, widening access beyond the infant population that has defined the therapy’s use since its original approval in 2019.
Apart from being a clinical milestone, the approval reopens the commercial narrative around Zolgensma, which has struggled to sustain early momentum despite its status as a breakthrough one-time therapy for a rare genetic disease.
Sales growth for Zolgensma slowed after the initial launch, reflecting both the small patient pool and competition from rival therapies. But analysts now see renewed upside in the newly eligible older population. Visible Alpha consensus forecasts show Zolgensma revenue rising 13% year-on-year in fiscal 2026 to $1.4 billion, with peak global sales of $2.1 billion projected by 2033, before an eventual decline as generic competition emerges.
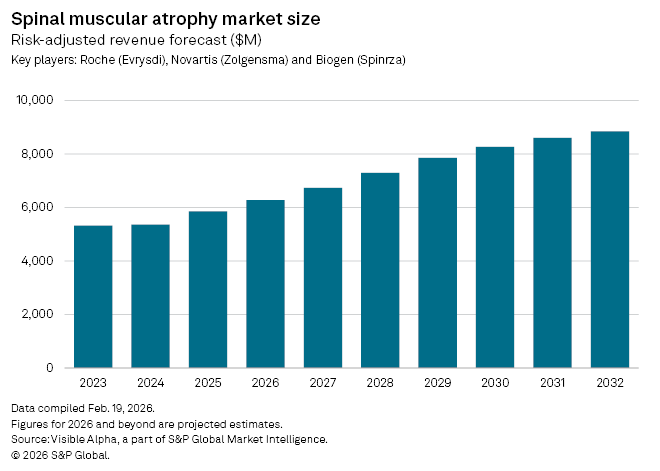
The broader SMA market is also expanding steadily. Visible Alpha estimates put global market size at $8.9 billion by 2032, up from $5.9 billion in 2025, implying a compound annual growth rate of just over 6%. Roche Holding AG (SIX: ROG) remains the market leader with Evrysdi, bolstered by an FDA-approved tablet formulation in February 2025, while Biogen (NASDAQ: BIIB) continues to defend its franchise with Spinraza, the first SMA therapy approved in 2016. Biogen is now advancing a higher-dose regimen, recently cleared in Japan and under review in Europe, though US regulators have requested additional data.
This article was published by Visible Alpha, part of S&P Global Market Intelligence and not by S&P Global Ratings, which is a separately managed division of S&P Global.
Content Type
Products & Offerings
Segment