Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Research — February 6, 2026
By Jay Rathod
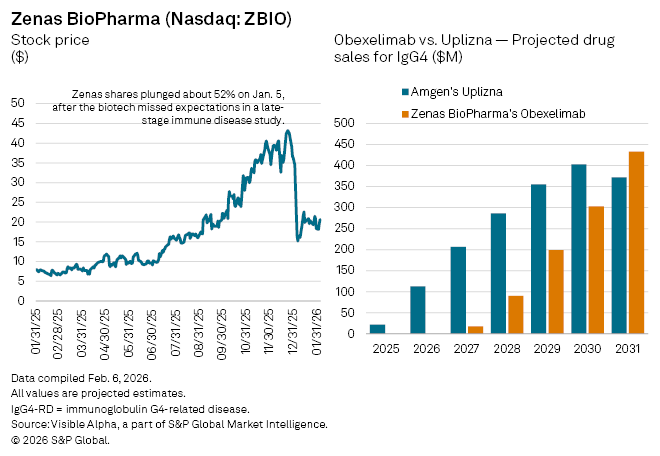
In early January, Zenas BioPharma (NASDAQ: ZBIO) announced that its phase III trial of obexelimab in immunoglobulin G4-related disease (IgG4) had met its primary endpoint. The shares of the clinical-stage biotech, however, fell 52% following the results, as the drug didn’t appear to match the effectiveness of the newly approved Uplizna from Amgen (NASDAQ: AMGN), the first approved treatment for IgG4-related disease, a rare autoimmune condition.
Even so, Visible Alpha consensus estimates suggest analysts remain constructive on obexelimab’s long-term revenue potential. Uplizna, approved in 2025 for IgG4-related disease, is projected to generate about $22 million in first-year sales in 2025, rising to $113 million in 2026 and reaching roughly $403 million by 2030.
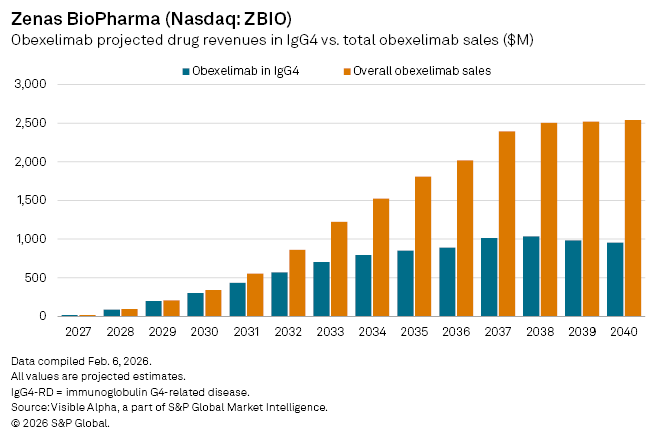
By comparison, obexelimab, which analysts expect could be approved in 2027, is forecast to deliver $18 million in risk-adjusted sales in its first year on the market. Revenues are then expected to accelerate sharply, climbing to $303 million by 2030 and about $851 million by 2035. Peak global sales of roughly $1.03 billion are penciled in for 2038, before erosion from generic competition sets in.
Analysts currently assign a probability of success of 53.33% for obexelimab in IgG4-related disease, highlighting both the uncertainty typical of late-stage biotech development and the competitive shadow cast by Amgen’s established therapy.
This article was published by Visible Alpha, part of S&P Global Market Intelligence and not by S&P Global Ratings, which is a separately managed division of S&P Global.
Content Type
Products & Offerings
Segment