Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Research — January 6, 2026
By Ankita Patil
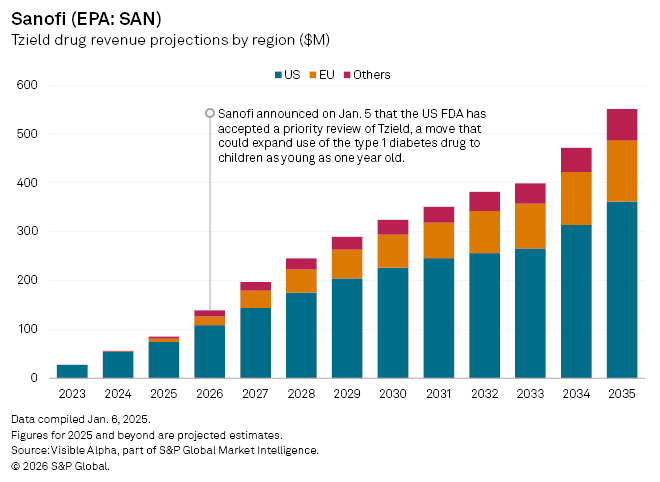
French pharmaceutical company Sanofi (EPA: SAN) has taken a fresh step to revive momentum for Tzield, its first-in-class therapy for type 1 diabetes, after the US FDA agreed to a priority review of an application to expand the drug’s use to younger children. The regulator is expected to deliver a decision by April 29.
Tzield is currently approved in the US to delay the onset of stage 3 type 1 diabetes in patients aged eight and older who have stage 2 diabetes. If approved, the new label would allow the drug to be used in children as young as one, materially enlarging the addressable population and potentially improving the drug’s long-term commercial outlook.
Tzield cleared by the FDA in November 2022, represents a novel preventive approach in an area of significant unmet need. Commercial traction, however, has so far been modest. Tzield generated $54 million in US sales in 2024. Outside the US, Tzield is only just beginning its international rollout, having recently been approved in the UK and China and recommended for approval in the EU.
Visible Alpha consensus points to a gradual ramp-up. Analysts expect US revenues of about $75 million in 2025, with a further $6 million from the EU and $4 million from other markets. Peak global sales are currently forecast at $579 million in 2035, before erosion from generic competition following patent expiry in 2034.
This article was published by Visible Alpha, part of S&P Global Market Intelligence and not by S&P Global Ratings, which is a separately managed division of S&P Global.
Content Type
Products & Offerings
Segment