Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Research — January 26, 2026
By Jueeli Kadam
Johnson & Johnson (NYSE: JNJ) and Legend Biotech Corp.’s (NASDAQ: LEGN) CAR-T therapy Carvykti has rapidly established itself as one of the best-selling treatments in relapsed or refractory multiple myeloma (RRMM), a notoriously difficult-to-treat blood cancer. Approved in 2022, Carvykti has benefited from strong efficacy data and broad uptake, helping to cement its position alongside Bristol-Myers Squibb Co.’s (NYSE: BMY) earlier entrant Abecma, which won approval in 2021.
But Carvykti’s commercial success comes with clinical caveats. Its label carries warnings for cytokine release syndrome and neurotoxicity, as well as an FDA-mandated boxed warning for rare but potentially severe immune-effector-cell-associated enterocolitis, safety considerations that can weigh on physician choice and patient eligibility.
That risk-benefit balance is now under closer scrutiny as Gilead Sciences Inc. (NASDAQ: GILD) advances a potential next-generation rival. Anito-cel (anitocabtagene autoleucel), developed by Gilead’s Kite unit in partnership with Arcellx, is in Phase 3 pivotal testing in the iMMagine-1 program for RRMM and is being positioned as a cleaner-profile alternative within the same BCMA-targeted CAR-T class.
In December, Gilead reported updated data from iMMagine-1 showing, notably, no cases of serious delayed neurological side effects or immune-related bowel inflammation, risks that have emerged with rival Carvykti weeks or months after treatment. The company is preparing anito-cel for regulatory filing, with a potential launch penciled in for 2026. Analysts currently assign a 67.2% probability of success to the therapy.
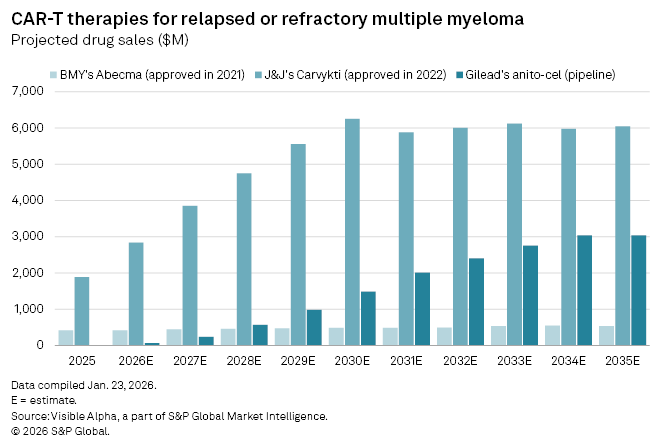
According to Visible Alpha consensus estimates, Carvykti is still expected to remain the dominant force in the category. Sales of the Johnson & Johnson–Legend therapy are projected to reach $2.8 billion in 2026, compared with $421 million for Bristol-Myers Squibb’s Abecma. Anito-cel, assuming approval in late 2026, is forecast to generate about $66 million in its first year on the market.
Longer term, however, analysts see greater upside for Gilead’s contender than for Abecma. Anito-cel is projected to deliver blockbuster revenue of $1.5 billion by 2030, rising to peak global sales of about $3 billion by 2034. Carvykti, by contrast, is expected to remain the category leader, with peak global sales estimated at $6.6 billion by 2030, while Abecma continues to lag both rivals over the forecast period.
This article was published by Visible Alpha, part of S&P Global Market Intelligence and not by S&P Global Ratings, which is a separately managed division of S&P Global.
Content Type
Products & Offerings
Segment