Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
S&P Global Offerings
Featured Topics
Featured Products
Events
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
Financial and Market intelligence
Fundamental & Alternative Datasets
Government & Defense
Professional Services
Banking & Capital Markets
Economy & Finance
Energy & Commodities
Technology & Innovation
Podcasts & Newsletters
11 Mar, 2026
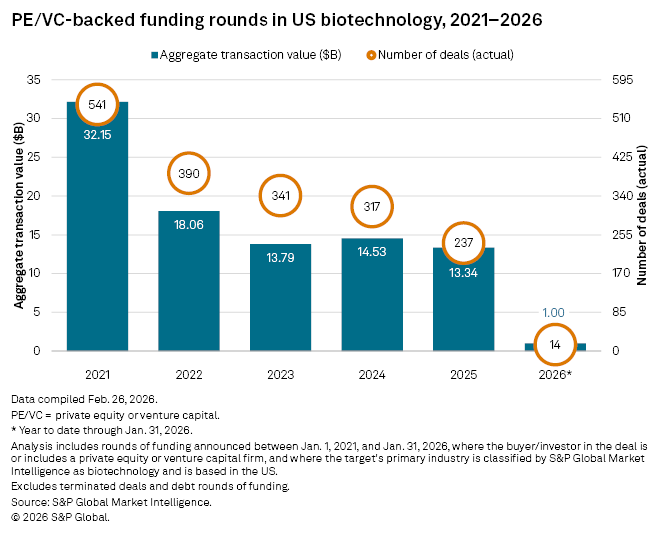
The number of venture capital-backed rounds of funding for US biotechnology companies fell by 25.2% year-over-year to 237, marking the lowest figure since at least 2021, according to S&P Global Market Intelligence data.
Transaction value also declined, albeit at a slower pace, dropping 8.19% year-over-year to $13.3 billion, the lowest value since at least 2021.
Policies at the federal level have impacted venture investment in biotech companies.
"Concerns around drug-pricing reforms, National Institute of Health funding pressure, and regulatory agency restructuring have increased perceived risk and lengthened investment timelines," said Rathanesh Ramasundram, head of Healthcare & Life Sciences, Advisory, at consulting firm Frost & Sullivan.
Additionally, high interest rates and a sluggish IPO biotech market have led investors to favor companies with established clinical validation, according to Aishvarya Agrawal, director of consulting firm YCP India.
"What you're seeing is more concentrated capital," he said. "Most of the funding is happening on Phase 2 assets and these are going to be less, but then the quantum of money that's going into them is going to be much larger."
A new drug from a biotech startup must pass through four phases before approval. Early phases carry the highest risk of failure, but if the drug eventually reaches approval, the early investors could reap very large multiples on their investment because they came in before most of the data on the drug existed.
However, only about 60% of drug candidates in the Phase 1 pre-testing stage advance to Phase 2, when the drug is tested on a small human population, Agrawal explained.
Phase 3 involves testing on a larger human population, carrying higher costs. Startups often partner with big pharmaceutical companies or raise late‑stage capital to run these trials.
"From a financing standpoint, [Phase 2 assets] offer credible clinical proof‑of‑concept and a clearer path to Phase 3, or partnership, which in turn improves exit options compared with purely Phase 1 assets," Agrawal said.
– Download a spreadsheet with data featured in this story.
– Explore the key trends shaping private equity-backed take-privates.
– Be updated on the latest private equity deals.
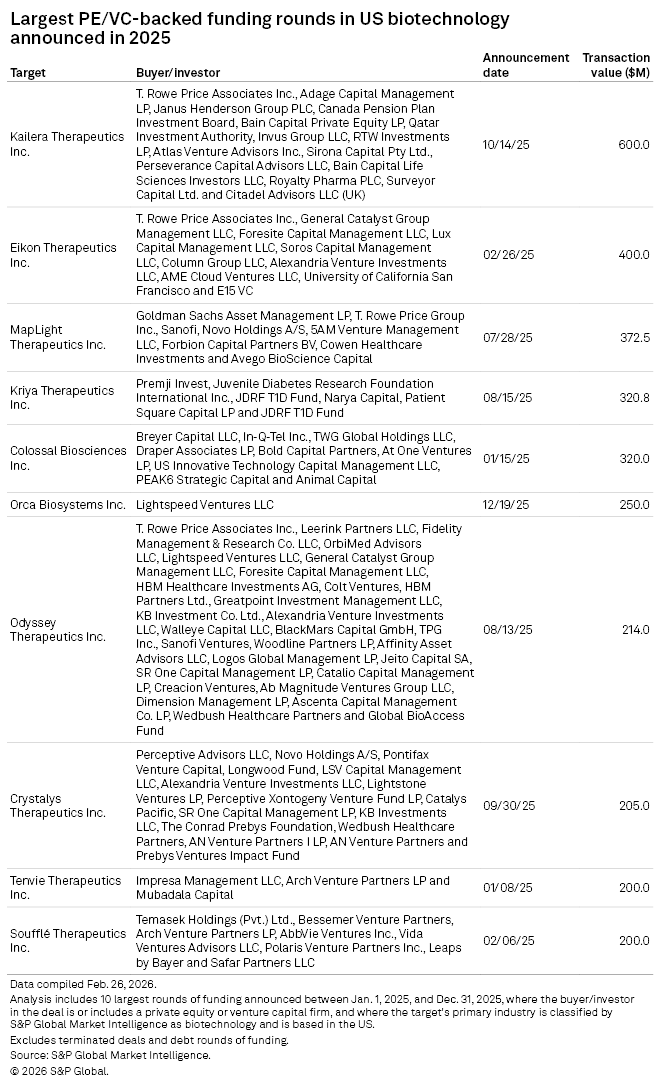
Largest deals
In February, Kailera Therapeutics Inc., which raised the largest round of funding for a US biotech company in 2025, totaling $600 million, concluded its Phase 2 trials for an obesity pill in China. It plans to initiate a global Phase 2 clinical trial within the year.
Eikon Therapeutics Inc., which secured the second-largest round for a US biotech in 2025, amounting to $400 million, is also conducting Phase 2 trials of its cancer immunotherapy drug.
AI-powered biotech
Capital is also flowing toward AI-enabled discovery platforms, according to Frost & Sullivan's Ramasundram.
Investors particularly value biotech firms with AI capabilities that can compress discovery timelines, improve hit rates or success probabilities and reduce development costs per program, Ramasundram said.
Currently, AI is mostly used to accelerate the early cycles of drug discovery. For example, China-based Insilico Medicine Inc. claims AI use cuts the pre-Phase 1 period, typically around 2.5 to 4.5 years, to 12 to 18 months.
"If AI‑enabled discovery continues to deliver shorter timelines and better success rates, it can structurally improve the risk–reward profile of biotech research and development, justifying higher capital allocation even in a more cautious market," Ramasundram said.
Biotech startups using AI have valuations twice as high as peer companies that are not using it, Agrawal added. AI-supported biotech startups are raising on average $80 million to $90 million, while others are raising $40 million to $50 million, he estimated.
Drug candidates discovered with AI support, however, have only reached Phase 2 testing, according to Agrawal. If the same drugs reach Phase 3, investment in biotechnology companies could increase.
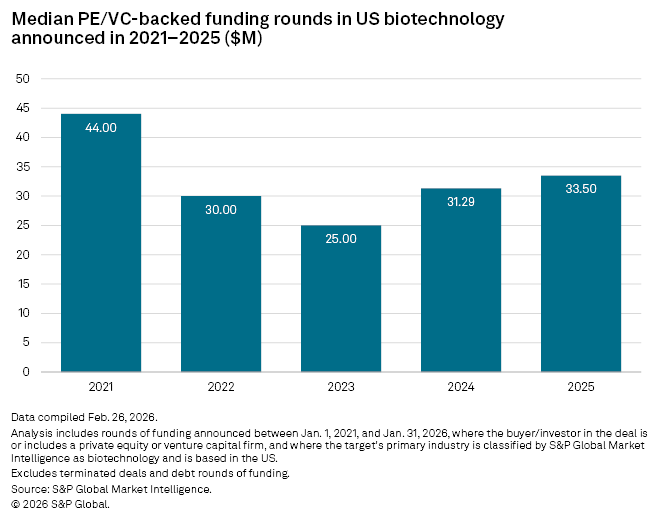
The promise of AI could be one factor behind the increase in median deal value in the sector, which rose 7% year on year to $33.5 million in 2025, Market Intelligence data shows.